Search any question & find its solution
Question:
Answered & Verified by Expert
A block of ice at \( -10^{\circ} \mathrm{C} \) slowly heated and converted to steam at \( 100^{\circ} \mathrm{C} \). Which of the following curves represents this phenomenon qualitatively?
Options:
Solution:
1706 Upvotes
Verified Answer
The correct answer is:
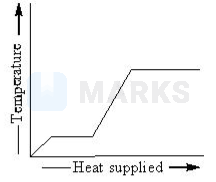

The temperature of ice will increases from .
Heat supplied in this process will be
Here, =mass of ice
=specific heat of ice
Then ice starts melting. Temperature during melting will remain constant (. Heat supplied in this process will be
Now, the temperature of water will increase from . Heat supplied will be
where .
Finally water at 100 will be converted into steam at 100 and during this process temperature again remains constant. Temperature heat supplied graph will be as follows.
Heat supplied in this process will be
Here, =mass of ice
=specific heat of ice
Then ice starts melting. Temperature during melting will remain constant (. Heat supplied in this process will be
Now, the temperature of water will increase from . Heat supplied will be
where .
Finally water at 100 will be converted into steam at 100 and during this process temperature again remains constant. Temperature heat supplied graph will be as follows.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.