Search any question & find its solution
Question:
Answered & Verified by Expert
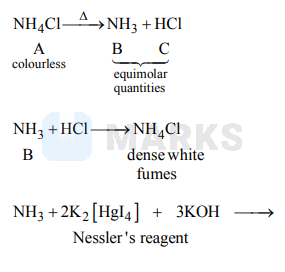
A colourless water soluble solid $A$ on heating gives equimolar quantities of $B$ and $C . B$ gives dense white fumes with $\mathrm{HCl}$ and $C$ does so with $\mathrm{NH}_{3} . B$ gives brown precipitate with Nessler's reagent and $C$ gives white precipitate with nitrates of $\mathrm{Ag}^{+}, \mathrm{Pb}^{+}$and $\mathrm{Hg}^{+} . A$ is
Options:
Solution:
2647 Upvotes
Verified Answer
The correct answer is:
$\mathrm{NH}_{4} \mathrm{Cl}$

Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.