Search any question & find its solution
Question:
Answered & Verified by Expert
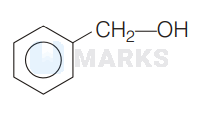
A compound ' $A^{\prime}\left(\mathrm{C}_{7} \mathrm{H}_{8} \mathrm{O}\right)$ is insoluble in $\mathrm{NaHCO}_{3}$ solution but dissolve in $\mathrm{NaOH}$ and give a characteristic colour with neutral $\mathrm{FeCl}_{3}$ solution. When treated with bromine water compound ' $A^{\prime}$ forms the compound $B$ with the formula $\mathrm{C}_{7} \mathrm{H}_{5} \mathrm{OBr}_{3} \cdot .^{\prime} A^{\prime}$ is
Options:
Solution:
1651 Upvotes
Verified Answer
The correct answer is:
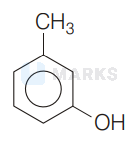

It is given in the question that, compound $A$ gives a characteristic colour with neutral $\mathrm{FeCl}_{3}$ solution and soluble in $\mathrm{NaOH}$ solution that means it contains a phenolic group.
Since compound ' $A$ ' when treated with $\mathrm{Br}_{2}$ forms compound ' $B$ ' with molecular formula $\mathrm{C}_{7} \mathrm{H}_{5} \mathrm{OBr}_{3}$ (ppt.).
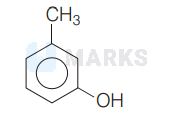
Considering the given information the given compound is most likely to be $m$-cresol. i.e.
Thus, the overall reactions are as follows.
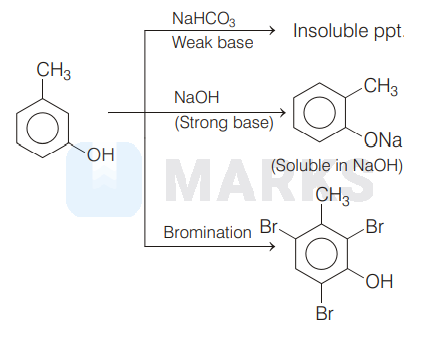
Since compound ' $A$ ' when treated with $\mathrm{Br}_{2}$ forms compound ' $B$ ' with molecular formula $\mathrm{C}_{7} \mathrm{H}_{5} \mathrm{OBr}_{3}$ (ppt.).
Considering the given information the given compound is most likely to be $m$-cresol. i.e.

Thus, the overall reactions are as follows.

Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.