Search any question & find its solution
Question:
Answered & Verified by Expert
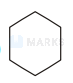
A compound $\mathrm{X}$ formed after heating coke with lime react with water to give $\mathrm{Y}$ which on passing over redhot iron at 873 produces $Z$. The compound $Z$ is
Options:
Solution:
2405 Upvotes
Verified Answer
The correct answer is:

$\begin{array}{l}
\mathrm{CaO}+\mathrm{C} \longrightarrow \mathrm{CaC}_{2}+\mathrm{CO}_{2} \\
\mathrm{CaC}_{2}+\mathrm{H}_{2} \mathrm{O} \longrightarrow \mathrm{HC} \equiv \mathrm{CH}+\mathrm{Ca}(\mathrm{OH})_{2} \\
3 \mathrm{HC} \equiv \mathrm{CH} \frac{\mathrm{Red} \mathrm{Hot}}{\mathrm{Fe}}
\end{array}$
\mathrm{CaO}+\mathrm{C} \longrightarrow \mathrm{CaC}_{2}+\mathrm{CO}_{2} \\
\mathrm{CaC}_{2}+\mathrm{H}_{2} \mathrm{O} \longrightarrow \mathrm{HC} \equiv \mathrm{CH}+\mathrm{Ca}(\mathrm{OH})_{2} \\
3 \mathrm{HC} \equiv \mathrm{CH} \frac{\mathrm{Red} \mathrm{Hot}}{\mathrm{Fe}}
\end{array}$
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.