Search any question & find its solution
Question:
Answered & Verified by Expert
A compound ' $\mathrm{X}^{\prime}$ upon reaction with $\mathrm{H}_2 \mathrm{O}$ produced a colourless gas ' $Y$ ' with rotten fish smell. Gas ' $\mathrm{Y}$ ' is absorbed in a solution of $\mathrm{CuSO}_4$ to give $\mathrm{Cu}_3 \mathrm{P}_2$ as one of the products. Predict the compound ' $\mathrm{X}$ '
Options:
Solution:
2871 Upvotes
Verified Answer
The correct answer is:
$\mathrm{Ca}_3 \mathrm{P}_2$
$$
\text { The given road map problem is }
$$
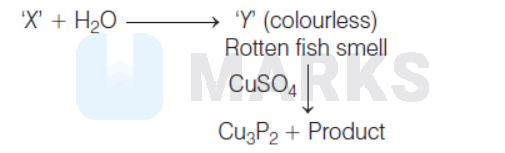
As compound ' $\mathrm{X}$ ' produces a colorless gas ' $\mathrm{Y}$ ' with rotten fish smell, it can be $\mathrm{Ca}_3 \mathrm{P}_2$ (calcium phosphide). The reaction takes palce as follows :
$$
\mathrm{Ca}_3 \mathrm{P}_2+\mathrm{H}_2 \mathrm{O} \longrightarrow \mathrm{Ca}(\mathrm{OH})_2+\mathrm{PH}_3
$$
Colorless gas with rotten fish smell. ' $\mathrm{Y}$ '
$\mathrm{CuSO}_4+\underset{\mathrm{Y}^{\prime}}{\mathrm{PH}_3} \longrightarrow \mathrm{Cu}_3 \mathrm{P}_2+\mathrm{H}_2 \mathrm{SO}_4$
\text { The given road map problem is }
$$

As compound ' $\mathrm{X}$ ' produces a colorless gas ' $\mathrm{Y}$ ' with rotten fish smell, it can be $\mathrm{Ca}_3 \mathrm{P}_2$ (calcium phosphide). The reaction takes palce as follows :
$$
\mathrm{Ca}_3 \mathrm{P}_2+\mathrm{H}_2 \mathrm{O} \longrightarrow \mathrm{Ca}(\mathrm{OH})_2+\mathrm{PH}_3
$$
Colorless gas with rotten fish smell. ' $\mathrm{Y}$ '
$\mathrm{CuSO}_4+\underset{\mathrm{Y}^{\prime}}{\mathrm{PH}_3} \longrightarrow \mathrm{Cu}_3 \mathrm{P}_2+\mathrm{H}_2 \mathrm{SO}_4$
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.