Search any question & find its solution
Question:
Answered & Verified by Expert
A container with rigid walls is covered with perfectly insulating material. The container is divided into two parts by a partition. One part contains a gas while the other is fully evacuated (vacuum). The partition is suddenly removed. The gas rushes to fill the entire volume and comes to equilibrium after a little time. If the gas is $\underline{\text { not }}$ ideal,
Options:
Solution:
2595 Upvotes
Verified Answer
The correct answer is:
the initial internal energy of the gas equals its final internal energy
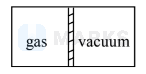
expansion is against
vacuum $\therefore \Delta \mathrm{W}=0$
Insulated container $\therefore \Delta \mathrm{Q}=0$
First law of thermodynamics
$\begin{array}{l}
\Delta \mathrm{Q}=\Delta \mathrm{W}+\Delta \mathrm{U} \\
0=0+\Delta \mathrm{U} \\
0=0+\Delta \mathrm{U} \\
\Delta \mathrm{U}=0
\end{array}$
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.