Search any question & find its solution
Question:
Answered & Verified by Expert
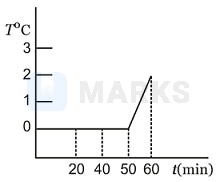
A cooking vessel on a slow burner contains 5 kg of water and an unknown mass of ice in equilibrium at 0oC at time t = 0. The temperature of the mixture is measured at various times and the result is plotted as shown in diagram. During the first 50 min the mixture remains at 0oC. From 50 min to 60 min, the temperature increases to 2oC. Neglecting the heat capacity of the heat capacity of the vessel, the initial mass of the ice is
Options:

Solution:
1063 Upvotes
Verified Answer
The correct answer is:
Let m be the mass of ice.
Rate of heat given by the burner is constant.
In the first 50 min
...(i)
From 50 min to 60 min
...(ii)
From Eqs. (i) and (ii)
Rate of heat given by the burner is constant.
In the first 50 min
...(i)
From 50 min to 60 min
...(ii)
From Eqs. (i) and (ii)
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.