Search any question & find its solution
Question:
Answered & Verified by Expert
A cubic structure is formed where atoms of element $X$ are occupied at corner of cube and also at face centers. Atoms of element $Y$ are present at body center and at the edge centers. If all the atoms are removed along a plane passing through the middle of the cube (bisecting the four edges), the formula will become
Options:
Solution:
2634 Upvotes
Verified Answer
The correct answer is:
$X Y$
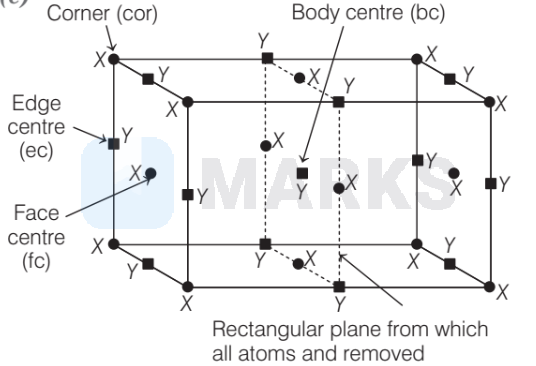
- $=X$, at corners (cor) and face centre (fc)
$\mathbf{-}=Y$, at edge centre $(\mathrm{ec})$ and body centre $(\mathrm{bc})$
After removal, number of atoms ratio in the crystal,
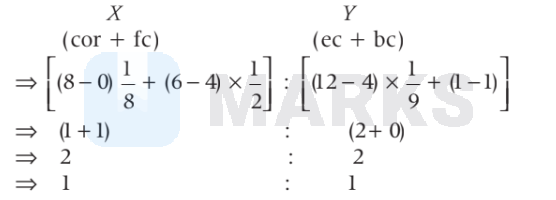
So, formula of the crystal will be $X Y$.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.