Search any question & find its solution
Question:
Answered & Verified by Expert
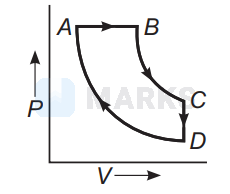
A cyclic process $A B C D$ is shown below in the given $P-V$ diagram. In the following answers the one that represents the same process as in $P-V$ diagram :
Options:

Solution:
2265 Upvotes
Verified Answer
The correct answer is:
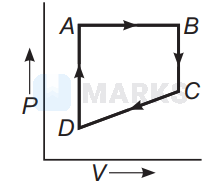

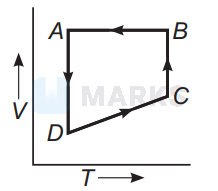
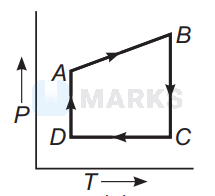
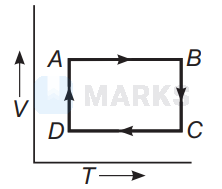
In $P$ - $V$ diagrams process $A B$ is isobaric process in which pressure remains constant i.e, $P=$ constant at all temperatures.
Process $B C$ is isothermal process in which, temperature remains constant i.e., $T=$ constant.
Process $C D$ is isochoric process in which volume remains constant i.e., $P-T$ diagram $C D$ is a straight line passing through origin.
Process $A D$ is adiabatic process which corresponds to process $A D$ in $P$ - $T$ diagram
Hence, the correct $P-T$ diagram is shown in option (a).
Process $B C$ is isothermal process in which, temperature remains constant i.e., $T=$ constant.
Process $C D$ is isochoric process in which volume remains constant i.e., $P-T$ diagram $C D$ is a straight line passing through origin.
Process $A D$ is adiabatic process which corresponds to process $A D$ in $P$ - $T$ diagram
Hence, the correct $P-T$ diagram is shown in option (a).
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.