Search any question & find its solution
Question:
Answered & Verified by Expert
A cylindrical adiabatic container of total volume is divided into two equal parts by a conducting piston which is free to move as shown in the figure. Each part contains identical gas at pressure . Initially the temperatures of the left and right parts are and respectively. An external force is applied on the piston of area to keep the piston at rest. The value of external force required when thermal equilibrium is reached is
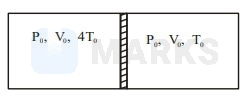
Solution:
1253 Upvotes
Verified Answer
The correct answer is:
If final temp is
Final pressure
In left
In right
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.