Search any question & find its solution
Question:
Answered & Verified by Expert
A cylindrical vessel of uniform cross-section consisting of a gas of $\gamma=1.5$ is divided into two parts $A$ and $B$ using a piston. Initially the piston is kept fixed such that part $A$ has pressure $p$ and volume $5 \mathrm{~V}$ and the part $B$ has pressure $8 p$ and volume $V$. If the piston is let free and the gas is allowed to undergo adiabatic process, then the final volume of the gas in part $A$ is
Options:
Solution:
2153 Upvotes
Verified Answer
The correct answer is:
$\frac{10}{3} v$
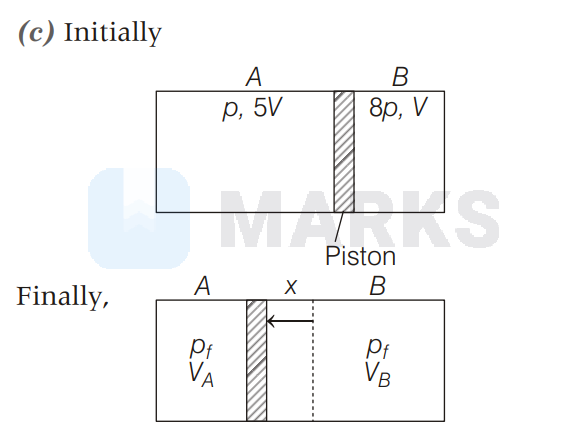
For adiabatic expression in part A and B, we have,
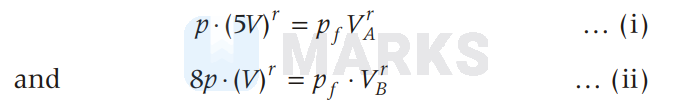
Solving for $V_A$ with $r=1 \cdot 5$, we get
$$
V_A=\frac{10}{3} \mathrm{~V}
$$
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.