Search any question & find its solution
Question:
Answered & Verified by Expert
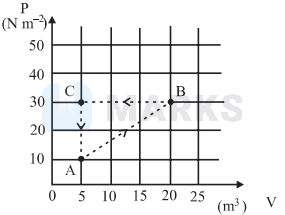
A gas in a closed container undergoes the cycle $\mathrm{ABCA}$ as shown in the figure. The net heat absorbed by the gas after it has completed 10 cycles is
Options:

Solution:
1028 Upvotes
Verified Answer
The correct answer is:
$-1.5 \mathrm{~kJ}$
$\Delta \mathrm{Q}=\Delta \mathrm{W} \quad[\because$ process is cyclic $]$
$=-\left[\frac{1}{2} \times 15 \times 20\right] \times 10 \quad[\because$ no. of cycles $=10]$
$=-1500 \mathrm{~J}$
As cycle is anti-clock wise. So $\Delta \mathrm{W}$ is -ve therefore, $\Delta \mathrm{Q}$ is -ve.
$=-\left[\frac{1}{2} \times 15 \times 20\right] \times 10 \quad[\because$ no. of cycles $=10]$
$=-1500 \mathrm{~J}$
As cycle is anti-clock wise. So $\Delta \mathrm{W}$ is -ve therefore, $\Delta \mathrm{Q}$ is -ve.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.