Search any question & find its solution
Question:
Answered & Verified by Expert
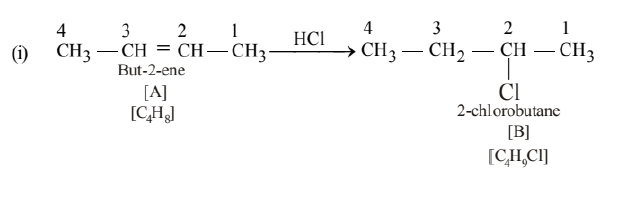
A hydrocarbon ' $\mathrm{A}$ ' $\left(\mathrm{C}_4 \mathrm{H}_8\right)$ on reaction with $\mathrm{HCl}$ gives a compound 'B', $\left(\mathrm{C}_4 \mathrm{H}_9 \mathrm{Cl}\right)$, which on reaction with $1 \mathrm{~mol}$ of $\mathrm{NH}_3$ gives compound ' $\mathrm{C}$ ', $\left(\mathrm{C}_4 \mathrm{H}_{11} \mathrm{~N}\right)$. On reacting with $\mathrm{NaNO}_2$ and $\mathrm{HCl}$ followed by treatment with water, compound ' $\mathrm{C}$ ' yields an optically active alcohol, 'D'. Ozonolysis of ' $A$ ' gives 2 mols of acetaldehyde. Identify compounds 'A' to 'D'. Explain the reactions involved.
Solution:
1085 Upvotes
Verified Answer
Compound 'A' is showing addition reaction with $\mathrm{HCl}$. Therefore it is an alkene. A on ozonolysis gives 2 moles of acetaldehyde. Therefore ' $\mathrm{A}$ ' is
$$
\mathrm{CH}_3-\mathrm{CH}=\mathrm{CH}-\mathrm{CH}_3 \text {. }
$$
Reactions involved are as following:



$$
\mathrm{CH}_3-\mathrm{CH}=\mathrm{CH}-\mathrm{CH}_3 \text {. }
$$
Reactions involved are as following:




Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.