Search any question & find its solution
Question:
Answered & Verified by Expert
A hydrogen atom moving at speed collides with another hydrogen atom kept at rest. Find the minimum value of for which one of the atoms may get ionised. The mass of a hydrogen atom Assume completely inelastic collision.
Options:
Solution:
2313 Upvotes
Verified Answer
The correct answer is:
Inelastic collision will take place, if a part of incident kinetic energy is utilised in exciting the atom. Here, one atom is to be ionised, i.e.
Assuming completely inelastic collision.
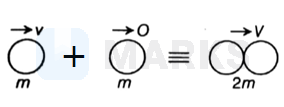
For an inelastic collision
By momentum conservation,
Assuming completely inelastic collision.
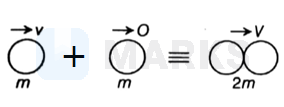
For an inelastic collision
By momentum conservation,
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.