Search any question & find its solution
Question:
Answered & Verified by Expert
A hydrogen-like atom (atomic number ) is in a higher excited state of quantum number . This excited atom can make a transition to the first excited state by successively emitting two photons of energies and respectively. Alternatively, the atom from the same excited state can make a transition to the second excited state by successively emitting two photons of energy and respectively. Determine the value of . [Ionization energy of hydrogen atom
Solution:
1371 Upvotes
Verified Answer
The correct answer is:
3
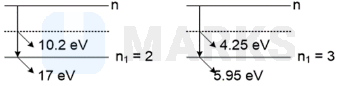
Solving the above two equation we get, and .
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.