Search any question & find its solution
Question:
Answered & Verified by Expert
A sample of HI (g) is placed in flask at a pressure of \(0.2\) atm. At equilibrium, the partial pressure of HI (g) is \(0.04\) atm. What is \(K_p\) for the given equilibrium ?
Solution:
2446 Upvotes
Verified Answer
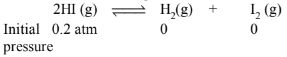
\(\begin{array}{lll}\text { At eqm, } 0.04 \mathrm{~atm} & 0.16 / 2 \mathrm{~atm} & 0.16 / 2 \mathrm{~atm} \\ & =0.08 \mathrm{~atm} & =0.08 \mathrm{~atm}\end{array}\)
(Decrease in the pressure of \(\mathrm{HI}=0.2-0.04=0.16 \mathrm{~atm}\).) \(\therefore \mathrm{K}_{\mathrm{p}}=\frac{\mathrm{p}_{\mathrm{H}_2} \times \mathrm{p}_{\mathrm{I}_2}}{\mathrm{p}_{\mathrm{HI}}^2}=\frac{0.08 \mathrm{~atm} \times 0.08 \mathrm{~atm}}{(0.04 \mathrm{~atm})^2}=4.0\)
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.