Search any question & find its solution
Question:
Answered & Verified by Expert
A solution containing components $\mathrm{A}$ and B follows Raoult's law if:
Options:
Solution:
2806 Upvotes
Verified Answer
The correct answer is:
attraction force remains same as in $\mathrm{A}-\mathrm{A}$ and $\mathrm{B}-\mathrm{B}$
Raoult's law is valid for ideal solution only. These two components A and B follows the condition of Raoult's law if the force of attraction between $\mathrm{A}$ and $\mathrm{B}$ is equal to the force of attraction between A-A and B-B
Positive non-ideal behavior of the vapor pressure of a solution follows Henry's Law at low concentrations and Raoult's Law at high concentrations (pure).
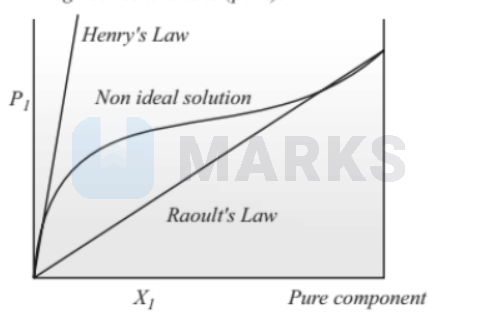
Positive non-ideal behavior of the vapor pressure of a solution follows Henry's Law at low concentrations and Raoult's Law at high concentrations (pure).

Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.