Search any question & find its solution
Question:
Answered & Verified by Expert
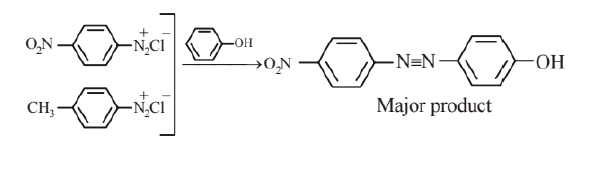
A solution contains $1 \mathrm{~g}$ mol. each of $\mathrm{p}$ - toluene diazonium chloride and $\mathrm{p}$ - nitrophenyl diazonium chloride. To this 1 $\mathrm{g}$ mol. of alkaline solution of phenol is added. Predict the major product. Explain your answer.
Solution:
1319 Upvotes
Verified Answer
$p$ - nitrophenyl diazonium chloride reacts preferentially with phenol. The given reaction is an electrophilic aromatic substitution reaction. Phenol in alkaline medium generates electron rich phenoxide ion. Since p-nitrophenyl diazonium chloride is stronger electrophile than p-toluene diazonium cation. Therefore it couples, preferentially with phenol. The reaction is given as follows

Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.