Search any question & find its solution
Question:
Answered & Verified by Expert
A solution when diluted with $\mathrm{H}_2 \mathrm{O}$ and boiled, it gives a white precipitate. On addition of excess $\mathrm{NH}_4 \mathrm{Cl} / \mathrm{NH}_4 \mathrm{OH}$, the volume of precipitate decreases leaving behind a white gelatinous precipitate. Identify the precipitate which dissolves in $\mathrm{NH}_4 \mathrm{OH} / \mathrm{NH}_4 \mathrm{Cl}$.
Options:
Solution:
2916 Upvotes
Verified Answer
The correct answer is:
$\mathrm{Zn}(\mathrm{OH})_2$
$\mathrm{Zn}(\mathrm{OH})_2$
A solution of $\mathrm{Zn}^{2+}$ ion reacts with water which on boiling give white ppt. of $\mathrm{Zn}(\mathrm{OH})_2$. This precipitates dissolve in excess of $\mathrm{NH}_4 \mathrm{OH}$ in presence of $\mathrm{NH}_4 \mathrm{Cl}$ because $\mathrm{Zn}(\mathrm{OH})_2$ on reaction with $\mathrm{NH}_4 \mathrm{OH}$ in presence of $\mathrm{NH}_4 \mathrm{Cl}$ forms tetrammine zinc (II) complex (soluble).
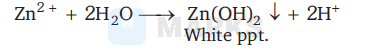
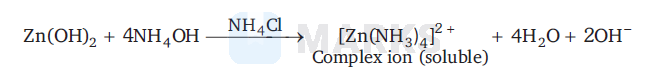
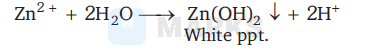
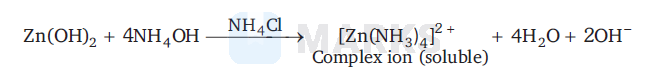
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.