Search any question & find its solution
A carbocation is a molecule in which a carbon atom has a positive charge and three bonds. We can say that, basically, they are carbon cations. Formerly, it was known as carbonium ion. Carbocation today is defined as any even-electron cation that possesses a significant positive charge on the carbon atom.
The carbon cations are very reactive and unstable due to an incomplete octet. In simple words, carbocations do not have eight electrons; therefore, they do not satisfy the octet rule.
In carbocation, the hybridisation of carbon will be and its shape is trigonal planar. There is also a vacant orbital which indicates its electron-deficient nature. The carbon has electrons in its valence shell. Due to this, it is an electron-deficient species, also known as an electrophile.
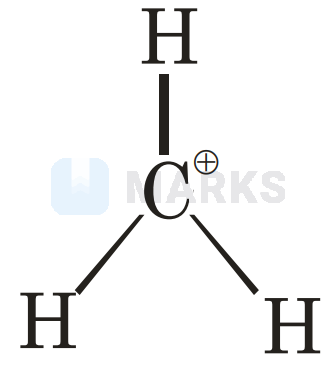
Six electron species
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.