Search any question & find its solution
Question:
Answered & Verified by Expert
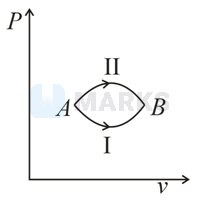
A system goes from to via two processes and shown in the figure. If and are the changes in internal energies in the processes I and Il respectively
Solution:
1900 Upvotes
Verified Answer
The correct answer is:
The process in which the initial and final state is the same is known as a cyclic process. It is a sequence of processes that leave the system in the same state in which it started.
Change in internal energy is independent on path, it depends on only initial and final state. So changes in internal energies for both process will be same.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.