Search any question & find its solution
Question:
Answered & Verified by Expert
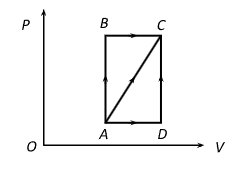
A thermodynamic process is shown in the figure. The pressures and volumes corresponding to some points in the figure are: $P_A=3 \times 10^4 \mathrm{~Pa}, P_B=8 \times 10^4 \mathrm{~Pa}$ and $V_A=2 \times 10^{-3} \mathrm{~m}^3, V_D=5 \times 10^{-3} \mathrm{~m}^3$ In process $A B, 600 \mathrm{~J}$ of heat is added to the system and in process $B C, 200 \mathrm{~J}$ of heat is added to the system. The change in internal energy of the system in process $A C$ would be
Options:

Solution:
1179 Upvotes
Verified Answer
The correct answer is:
$560 \mathrm{~J}$
By adjoining graph $W_{A B}=0$ and
$\begin{gathered}W_{B C}=8 \times 10^4[5-2] \times 10^{-3}=240 \mathrm{~J} \\ \therefore W_{A C}=W_{A B}+W_{B C}=0+240=240 \mathrm{~J}\end{gathered}$
Now, $\Delta Q_{A C}=\Delta Q_{A B}+\Delta Q_{B C}=600+200=800 \mathrm{~J}$
From FLOT $\Delta Q_{A C}=\Delta U_{A C}+\Delta W_{A C}$
$\Rightarrow 800=\Delta U_{A C}+240 \Rightarrow \Delta U_{A C}=560 \mathrm{~J}$
$\begin{gathered}W_{B C}=8 \times 10^4[5-2] \times 10^{-3}=240 \mathrm{~J} \\ \therefore W_{A C}=W_{A B}+W_{B C}=0+240=240 \mathrm{~J}\end{gathered}$
Now, $\Delta Q_{A C}=\Delta Q_{A B}+\Delta Q_{B C}=600+200=800 \mathrm{~J}$
From FLOT $\Delta Q_{A C}=\Delta U_{A C}+\Delta W_{A C}$
$\Rightarrow 800=\Delta U_{A C}+240 \Rightarrow \Delta U_{A C}=560 \mathrm{~J}$
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.