Search any question & find its solution
Question:
Answered & Verified by Expert
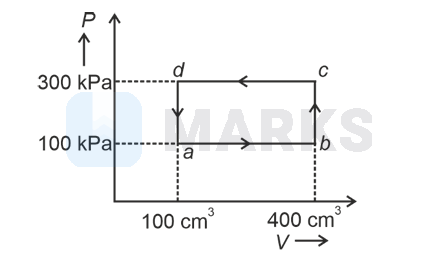
A thermodynamic system is taken through the cycle $a b c d a$. The work done by the gas along the path $b c$ is:
Options:

Solution:
1656 Upvotes
Verified Answer
The correct answer is:
Zero
Path $b c$ is an isochoric process.
$\therefore \quad$ Work done by gas along path $b c$ is zero.
$\therefore \quad$ Work done by gas along path $b c$ is zero.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.