Search any question & find its solution
Question:
Answered & Verified by Expert
A thermodynamic system is taken through the cycle as shown in the figure. Heat rejected by the gas during the cycle is
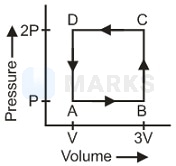
Options:

Solution:
2715 Upvotes
Verified Answer
The correct answer is:
In a cyclic process . In a cyclic process work done is equal to the area under the cycle and is positive if the cycle is clockwise and negative if anticlockwise.
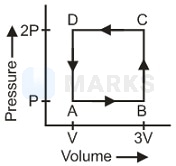
According to the first law of thermodynamics
or
i.e., heat supplied to the system is equal to the work done
So heat absorbed,
Heat rejected by the gas =

According to the first law of thermodynamics
or
i.e., heat supplied to the system is equal to the work done
So heat absorbed,
Heat rejected by the gas =
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.