Search any question & find its solution
Question:
Answered & Verified by Expert
Acidity of \(\mathrm{BF}_3\) can be explained on the basis of which of the following concepts?
Options:
Solution:
2213 Upvotes
Verified Answer
The correct answer is:
Lewis concept
\(\mathrm{B}\) is \(\mathrm{BF}_3\) is electron deficient i.e, lone-pair of electrons acceptor (Lewis acid), because in \(\mathrm{BF}_3\), \(B\) is \(s p^2\) hybridised and it does not satisfy octet ( \(\mathrm{B} \Rightarrow\) has say bonded electrons).
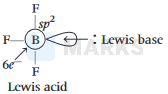
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.