Search any question & find its solution
Question:
Answered & Verified by Expert
Action of soap is due to emulsification and micelle formation. Comment.
Solution:
2263 Upvotes
Verified Answer
Soap is sodium or potassium salt of a higher fatty acid and may be represented as \(\mathrm{RCOO}^{-} \mathrm{Na}^{+}\)(e.g., sodium stearate \(\mathrm{CH}_3\left(\mathrm{CH}_2\right)_{16} \mathrm{COO}^{-} \mathrm{Na}^{+}\), which is a major component of many bar soaps). When dissolved in water, it dissociates into \(R \mathrm{COO}^{-}\)and \(\mathrm{Na}^{+}\)ions. The \(\mathrm{RCOO}^{-}\)ions, however, consist of two parts - a long hydrocarbon chain \(R\) (also called non-polar 'tail') which is hydrophobic (water repelling), and a polar group \(\mathrm{COO}^{-}\)(also called polar- ionic 'head'), which is hydrophilic (water loving).
The \(\mathrm{RCOO}^{-}\)ions are, therefore, present on the surface with their \(\mathrm{COO}^{-}\)groups in water and the hydrocarbon chains \(R\) staying away from it and remain at the surface. But at critical micelle concentration, the anions are pulled into the bulk of the solution and aggregate to form a spherical shape with their hydrocarbon chains pointing towards the centre of the sphere with \(\mathrm{COO}^{-}\)part remaining outward on the surface of the sphere. An aggregate thus formed is known as 'ionic micelle'.
The cleansing action of soap is due to the fact that soap molecules form micelle around the oil droplet in such a way that hydrophobic part of the stearate ions is in the oil droplet and hydrophilic part projects out of the grease droplet like the bristles. Since the polar groups can interact with water, the oil droplet surrounded by stearate ions is now pulled in water and removed from the dirty surface. Thus soap helps in emulsification and washing away of oils and fats. The negatively charged sheath around the globules prevents them from coming together and forming aggregates.
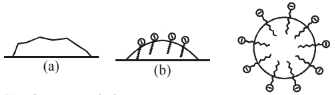
(a) Grease on cloth
(b) Stearate ions (from soap) arranging around the grease droplets
(c) Micelle formed
The \(\mathrm{RCOO}^{-}\)ions are, therefore, present on the surface with their \(\mathrm{COO}^{-}\)groups in water and the hydrocarbon chains \(R\) staying away from it and remain at the surface. But at critical micelle concentration, the anions are pulled into the bulk of the solution and aggregate to form a spherical shape with their hydrocarbon chains pointing towards the centre of the sphere with \(\mathrm{COO}^{-}\)part remaining outward on the surface of the sphere. An aggregate thus formed is known as 'ionic micelle'.
The cleansing action of soap is due to the fact that soap molecules form micelle around the oil droplet in such a way that hydrophobic part of the stearate ions is in the oil droplet and hydrophilic part projects out of the grease droplet like the bristles. Since the polar groups can interact with water, the oil droplet surrounded by stearate ions is now pulled in water and removed from the dirty surface. Thus soap helps in emulsification and washing away of oils and fats. The negatively charged sheath around the globules prevents them from coming together and forming aggregates.

(a) Grease on cloth
(b) Stearate ions (from soap) arranging around the grease droplets
(c) Micelle formed
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.