Search any question & find its solution
Question:
Answered & Verified by Expert
$\mathrm{AgNO}_3(a q)$ was added to an aqueous $\mathrm{KCl}$ solution gradually and the conductivity of the solution was measured. The plot of conductance $(\Lambda)$ versus the volume of $\mathrm{AgNO}_3$ is
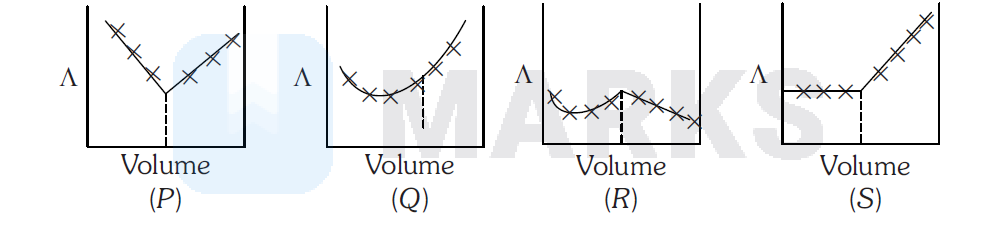
Options:

Solution:
1761 Upvotes
Verified Answer
The correct answer is:
$S$
$S$
As $\mathrm{AgNO}_3$ is added to solution, $\mathrm{KCl}$ will be displaced accrding to following reaction
$$
\mathrm{AgNO}_3(a q)+\mathrm{KCl}(a q) \longrightarrow \operatorname{AgCl}(s)+\mathrm{KNO}_3(a q)
$$
For every mole of $\mathrm{KCl}$ displaced from solution, one mole of $\mathrm{KNO}_3$ comes in solution resulting in almost constant conductivity. As the end point is reached, added $\mathrm{AgNO}_3$ remain in solution increasing ionic concentration, hence conductivity increases.
$$
\mathrm{AgNO}_3(a q)+\mathrm{KCl}(a q) \longrightarrow \operatorname{AgCl}(s)+\mathrm{KNO}_3(a q)
$$
For every mole of $\mathrm{KCl}$ displaced from solution, one mole of $\mathrm{KNO}_3$ comes in solution resulting in almost constant conductivity. As the end point is reached, added $\mathrm{AgNO}_3$ remain in solution increasing ionic concentration, hence conductivity increases.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.