Search any question & find its solution
Question:
Answered & Verified by Expert
Among 2-chloropropanoic acid,
3-chloro-propanoic acid, 2-2-dichloroacetic acid and propanoic acid the $K_{a}$ values will be in the order,
Options:
3-chloro-propanoic acid, 2-2-dichloroacetic acid and propanoic acid the $K_{a}$ values will be in the order,
Solution:
1949 Upvotes
Verified Answer
The correct answer is:
2 , 2-dichloroacetic acid $>2$-chloropropanoic acid $>3$-chloropropanoic acid $>$ propanoic acid
Acidic strength $\propto k_{a}$ values
Due to - I-effect of electron withdrawing - Cl group, chloropropanoic acid is stronger acid than propanoic acid. Further, greater the number of electron withdrawing substitutents, greater would be the acidic strength.
Inductive effect decreases rapidly with distance and so, is the acidic strength. Hence, the correct order of acidic strength (or $k_{a}$ values) will be
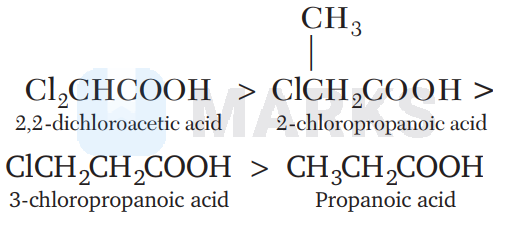
Due to - I-effect of electron withdrawing - Cl group, chloropropanoic acid is stronger acid than propanoic acid. Further, greater the number of electron withdrawing substitutents, greater would be the acidic strength.
Inductive effect decreases rapidly with distance and so, is the acidic strength. Hence, the correct order of acidic strength (or $k_{a}$ values) will be

Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.