Search any question & find its solution
Question:
Answered & Verified by Expert
Among the complexes of Ni listed below, the following is the only correct statement about the given compounds.
I. $\left[\mathrm{Ni}(\mathrm{CO})_{4}\right]$
II. $\left[\mathrm{Ni}(\mathrm{CN})_{4}\right]^{2-}$
III. $\left[\mathrm{NiCl}_{4}\right]^{2-}$
Options:
I. $\left[\mathrm{Ni}(\mathrm{CO})_{4}\right]$
II. $\left[\mathrm{Ni}(\mathrm{CN})_{4}\right]^{2-}$
III. $\left[\mathrm{NiCl}_{4}\right]^{2-}$
Solution:
1034 Upvotes
Verified Answer
The correct answer is:
I and III are tetrahedral and II has square planar geometry.
${ }_{28} \mathrm{Ni}=3 d^{8} 4 s^{2}$
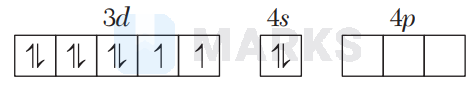
In $\left[\mathrm{Ni}(\mathrm{CO})_{4}\right], \mathrm{CO}$ is a strong field ligand which causes pairing of electrons.
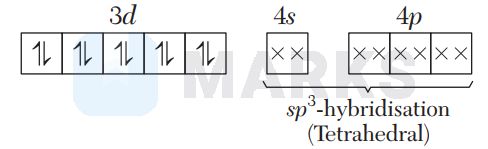
In $\left[\mathrm{Ni}(\mathrm{CN})_{4}\right]^{2-}, \mathrm{CN}^{-}$is a strong field ligand which causes pairing of electrons.
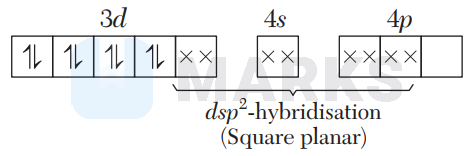
In $\left[\mathrm{NiCl}_{4}\right]^{2-}, \mathrm{Cl}^{-}$is a weak field ligand, so no pairing of electrons takes place.
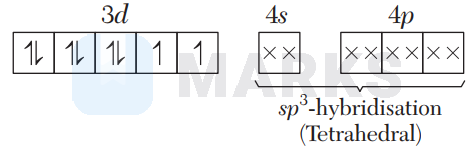
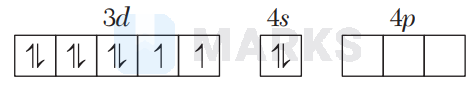
In $\left[\mathrm{Ni}(\mathrm{CO})_{4}\right], \mathrm{CO}$ is a strong field ligand which causes pairing of electrons.

In $\left[\mathrm{Ni}(\mathrm{CN})_{4}\right]^{2-}, \mathrm{CN}^{-}$is a strong field ligand which causes pairing of electrons.

In $\left[\mathrm{NiCl}_{4}\right]^{2-}, \mathrm{Cl}^{-}$is a weak field ligand, so no pairing of electrons takes place.

Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.