Search any question & find its solution
Question:
Answered & Verified by Expert
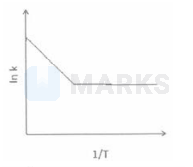
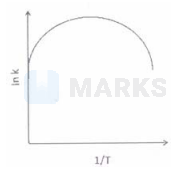
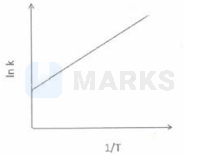
Among the following graphs showing variation of rate $(\mathrm{k})$ with temperature (T) for a reaction, the one that exhibits arrhenius behavior over the entire temperature range is -
Options:
Solution:
1836 Upvotes
Verified Answer
The correct answer is:
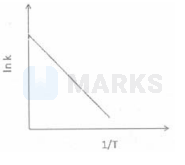

$\begin{array}{l}
\mathrm{K}=\mathrm{Ae}^{-} \frac{\mathrm{E}_{\mathrm{a}}}{\mathrm{RT}} \\
\ln \mathrm{K}=\ln \mathrm{A}-\frac{\mathrm{E}_{\mathrm{a}}}{\mathrm{RT}}
\end{array}$
\mathrm{K}=\mathrm{Ae}^{-} \frac{\mathrm{E}_{\mathrm{a}}}{\mathrm{RT}} \\
\ln \mathrm{K}=\ln \mathrm{A}-\frac{\mathrm{E}_{\mathrm{a}}}{\mathrm{RT}}
\end{array}$
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.