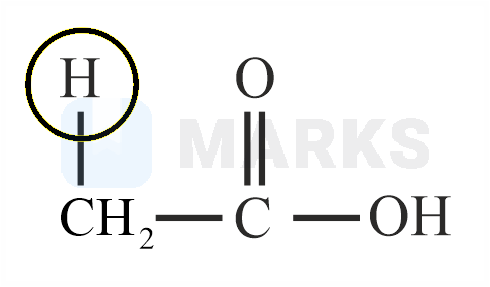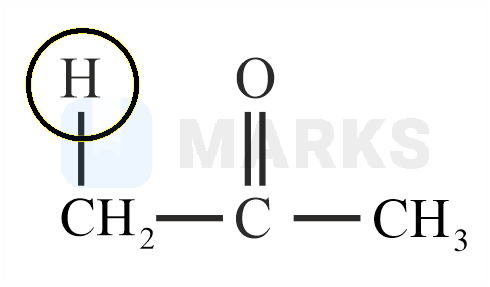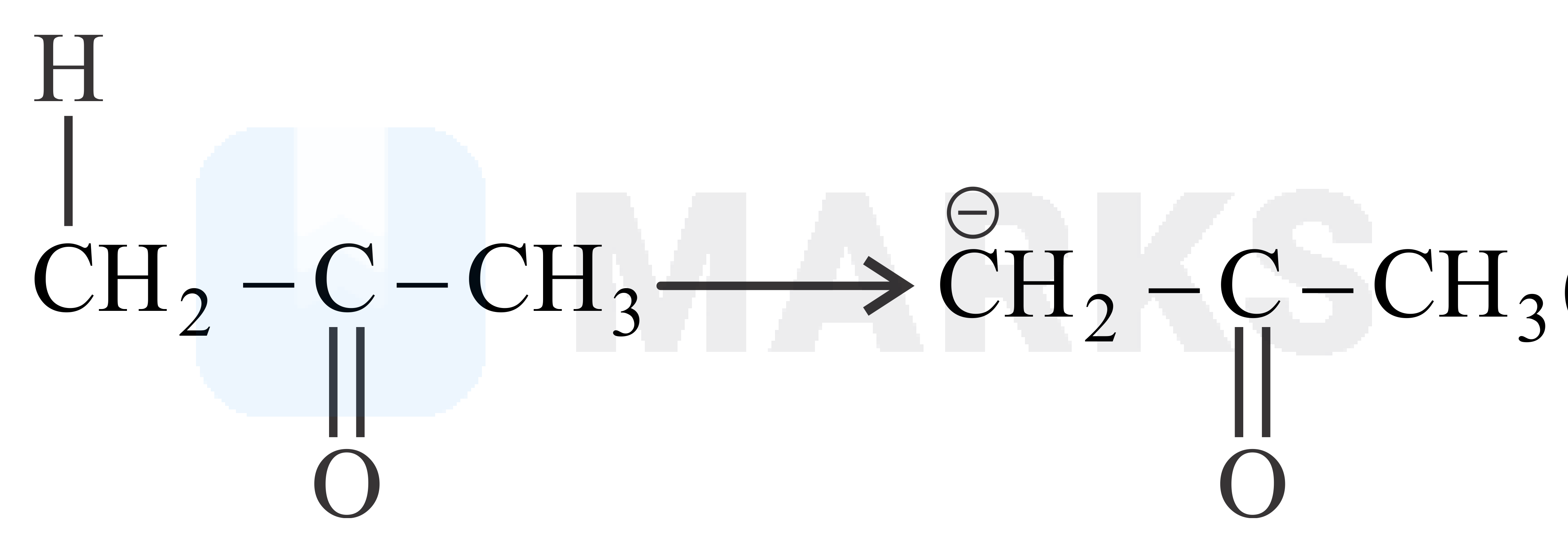Search any question & find its solution
Question:
Answered & Verified by Expert
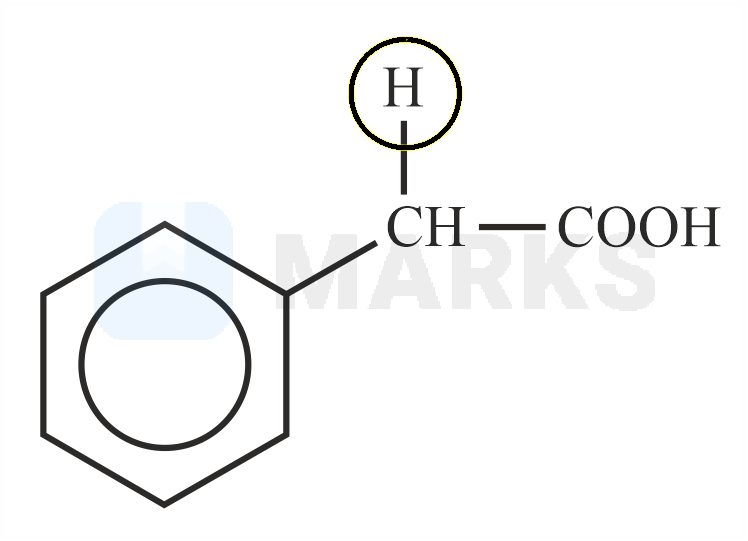
Among the following marked proton of which compound shows lowest value?
Options:
Solution:
1641 Upvotes
Verified Answer
The correct answer is:
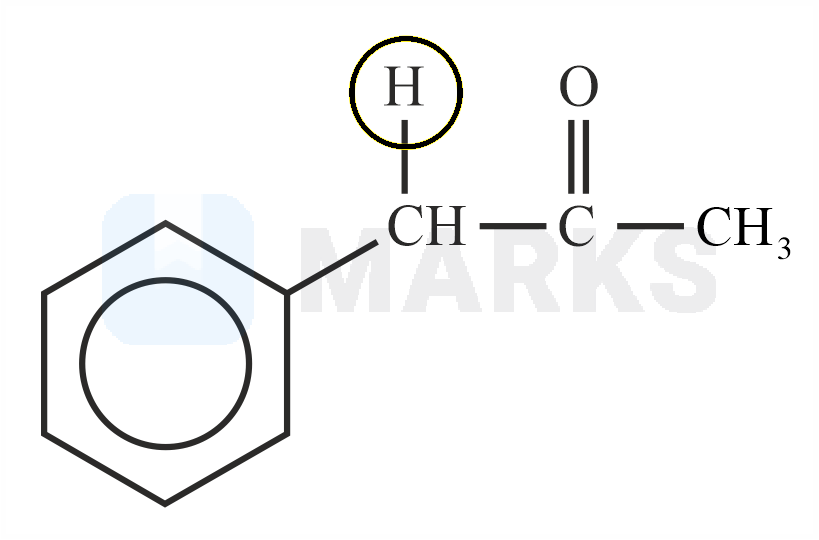
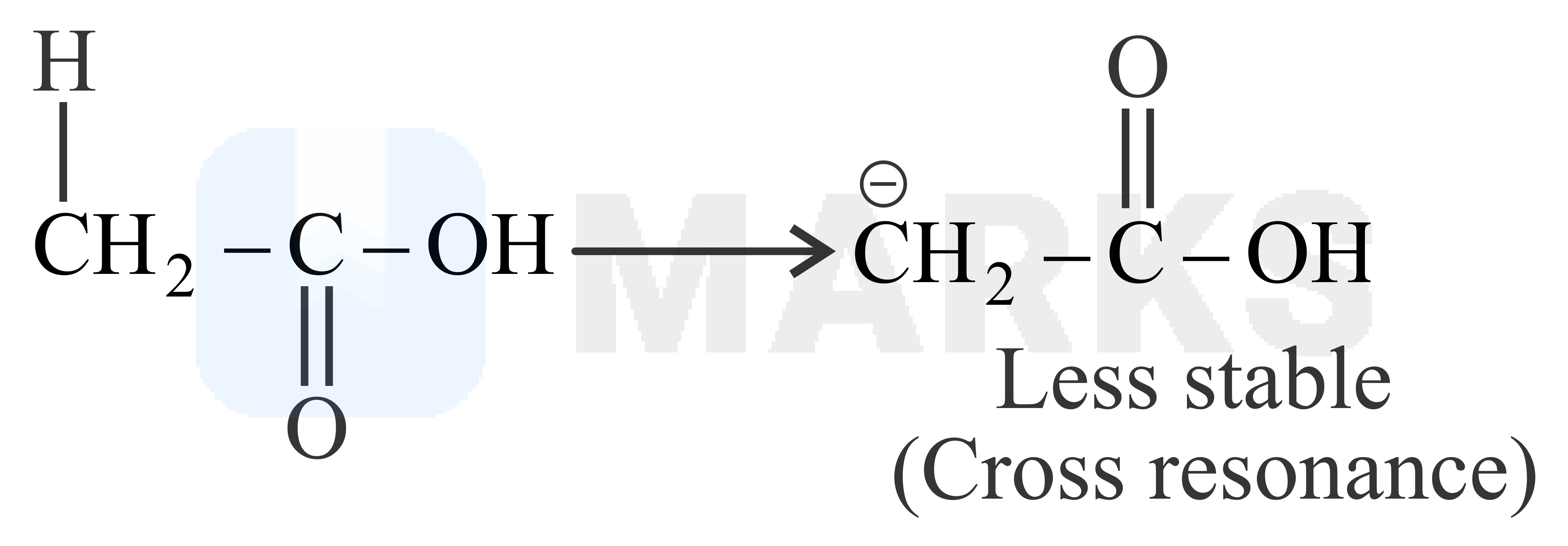
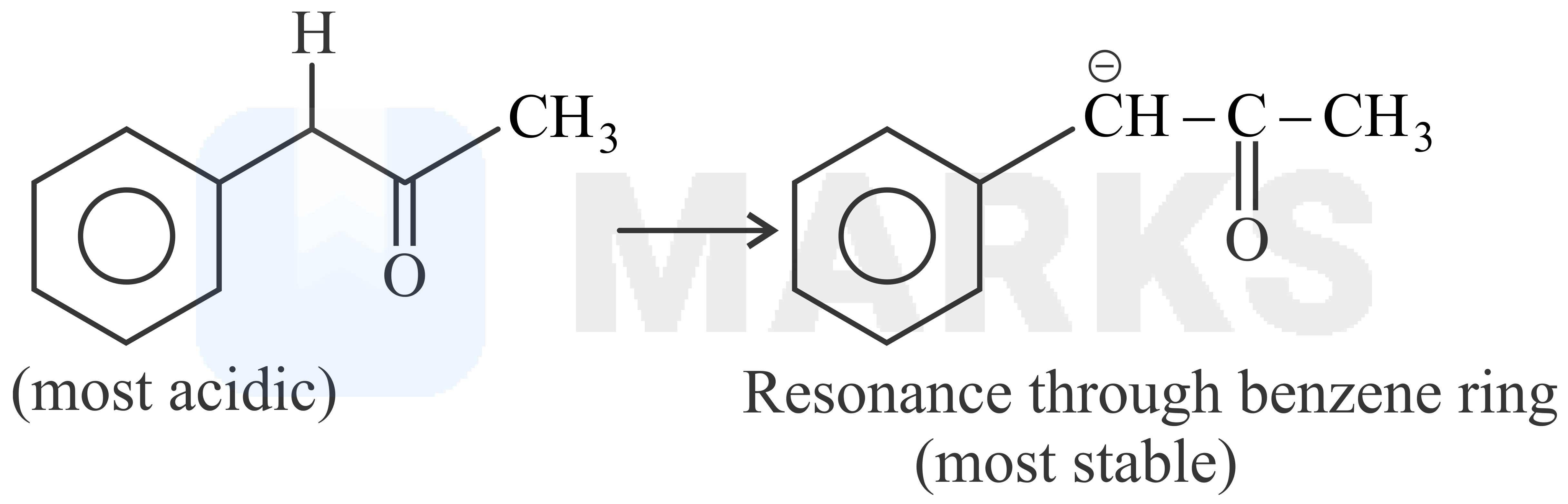
The smaller the value of , the stronger the acid. The acidic nature of the compound depends on the stability of conjugate base.
The stability of conjugate base formed by above compound is more stable than other three compounds given.
So it has least value.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.