Search any question & find its solution
Question:
Answered & Verified by Expert
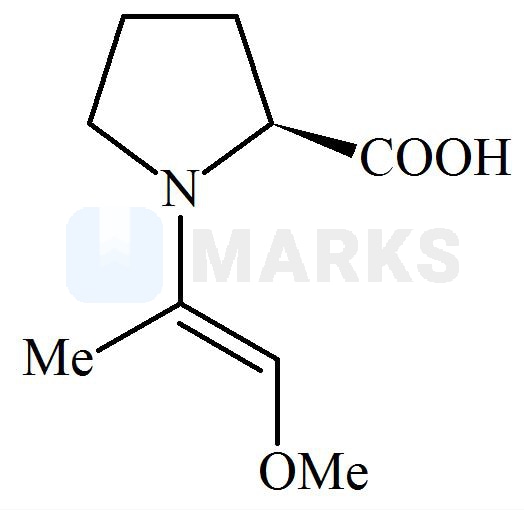
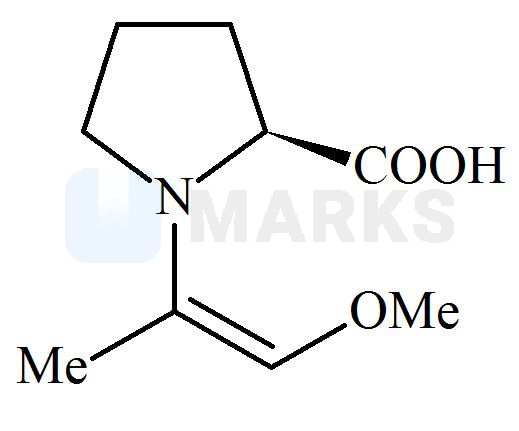
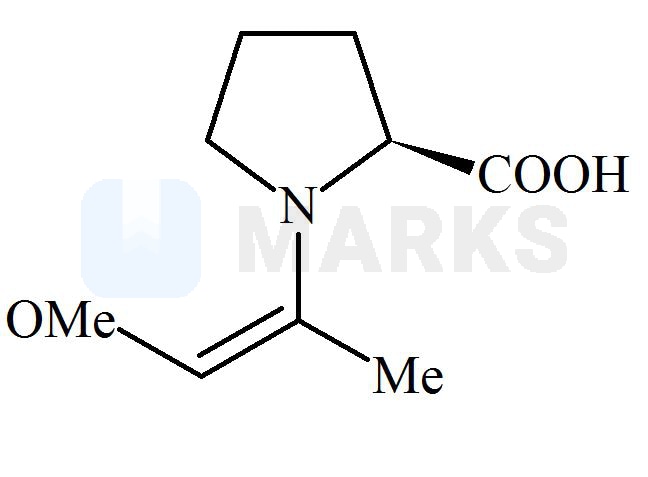
Among the following structures, which will show the most stable enamine formation?
(Where is )
Solution:
1491 Upvotes
Verified Answer
The correct answer is:
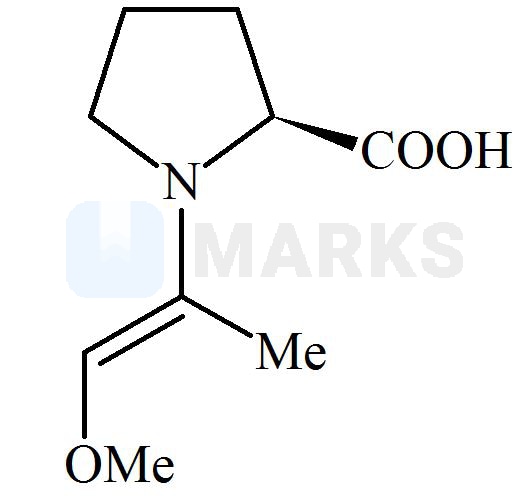

All these enamines are interconvertible through their resonating structures. So most stable form is ‘C’ due to steric factor. All of these has the equal resonance only differ in their geometrical configuration. The enamine in option C has bulky groups on opposite side of double bond. means they have the minimum repulsion between them.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.