Search any question & find its solution
Question:
Answered & Verified by Expert
An alkene $\mathrm{X}$ with formula $\mathrm{C}_4 \mathrm{H}_8$ does not exhibit geometrical isomerism. In the conversion of $\mathrm{X}$ to $\mathrm{Y}$, the correct sequence of reagents A and B used are ( $\mathrm{Y}$. gives iodoform test)
Options:
Solution:
2527 Upvotes
Verified Answer
The correct answer is:
$\mathrm{H}_2 \mathrm{O} / \mathrm{H}^{+}, \quad \mathrm{Cu} / 573 \mathrm{~K}$
Since ' $\mathrm{X}$ ' does not exhibit geometrical isomerism, two groups on the doubly-bonded carbon atoms will be identical for each carbon.
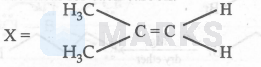
$$
\text { or } \mathrm{CH}_3 \mathrm{CH}_2 \mathrm{CH}=\mathrm{CH}_2
$$
$\mathrm{Y}=$ Gives iodoform test so it must be a methyl alcohol or methyl ketone.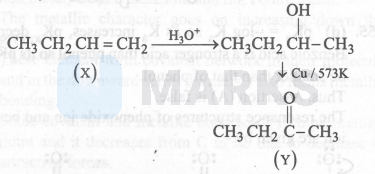
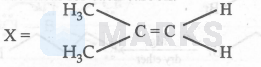
$$
\text { or } \mathrm{CH}_3 \mathrm{CH}_2 \mathrm{CH}=\mathrm{CH}_2
$$
$\mathrm{Y}=$ Gives iodoform test so it must be a methyl alcohol or methyl ketone.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.