Search any question & find its solution
Question:
Answered & Verified by Expert
An amine \((\mathrm{X})\) is prepared by ammonolysis of benzyl chloride. On adding p-toluenesulphonyl chloride to it the solution remains clear. Molar mass of the amine (X) formed is ______ \(\mathrm{g} \mathrm{mol}^{-1}\).
(Given molar mass in \(\mathrm{gmol}^{-1} \mathrm{C}: 12, \mathrm{H}: 1, \mathrm{O}: 16, \mathrm{~N}: 14\))
(Given molar mass in \(\mathrm{gmol}^{-1} \mathrm{C}: 12, \mathrm{H}: 1, \mathrm{O}: 16, \mathrm{~N}: 14\))
Solution:
1233 Upvotes
Verified Answer
The correct answer is:
287
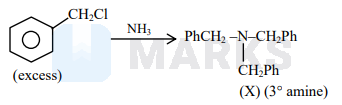
Molar Mass of $(\mathrm{X})$ is $287 \mathrm{~g} \mathrm{~mol}^{-1}$
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.