Search any question & find its solution
Question:
Answered & Verified by Expert
An amount of solid $\mathrm{NH}_4 \mathrm{HS}$ is placed in a flask already containing ammonia gas at a certain temperature and $0.50 \mathrm{~atm}$. Pressure. Ammonium hydrogen sulphide decomposes to yield $\mathrm{NH}_3$ and $\mathrm{H}_2 \mathrm{~S}$ gases in the flask. When the decomposition reaction reaches equilibrium, the total pressure in the flask rises to $0.84 \mathrm{~atm}$. The equilibrium constant for $\mathrm{NH}_4 \mathrm{HS}$ decomposition at this temperature is
Options:
Solution:
2426 Upvotes
Verified Answer
The correct answer is:
$0.11$
$0.11$
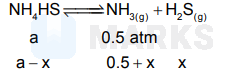
Total pressure $=0.5+2 \mathrm{x}=0.84$
i.e., $\mathrm{x}=0.17$
$\mathrm{~K}_{\mathrm{p}}=\mathrm{p}_{\mathrm{NH}_3} \cdot \mathrm{p}_{\mathrm{H}_2 \mathrm{~s}}$
$=(0.67) .(0.17)$
$\quad=0.1139$.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.