Search any question & find its solution
Question:
Answered & Verified by Expert
An aromatic compound 'A' (molecular formula $\mathrm{C}_8 \mathrm{H}_8 \mathrm{O}$ ) gives positive 2, 4-DNP test. It gives a yellow precipitate of compound ' $\mathrm{B}$ ' on treatment with iodine and sodium hydroxide solution. Compound 'A' does not give Tollen's or Fehling's test. On drastic oxidation with potassium permanganate it forms a carboxylic acid ' $\mathrm{C}$ ' (molecular formula $\mathrm{C}_7 \mathrm{H}_6 \mathrm{O}_2$ ), which is also formed along with the yellow compound in the above reaction. Identify $A, B$ and $\mathrm{C}$ and write all the reactions involved.
Solution:
1152 Upvotes
Verified Answer
Molecular formula of given aromatic compound is $\mathrm{C}_8 \mathrm{H}_8 \mathrm{O}$. Possible structures of aromatic compound with molecular formula $\mathrm{C}_8 \mathrm{H}_8 \mathrm{O}$ are as follows.
(i)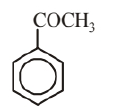
(ii)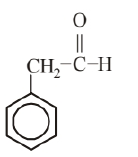
(iii)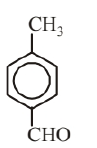
(iv)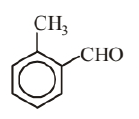
(v)
Since, 'A' does not give Tollen's and Fehling's test. Therefore it should be a ketone and not aldeh yde. So, (i) is the correct structure. The reactions involved are as following: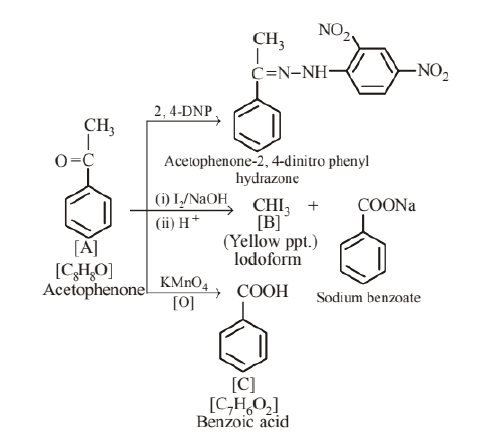
(i)

(ii)

(iii)

(iv)

(v)

Since, 'A' does not give Tollen's and Fehling's test. Therefore it should be a ketone and not aldeh yde. So, (i) is the correct structure. The reactions involved are as following:

Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.