Search any question & find its solution
Question:
Answered & Verified by Expert
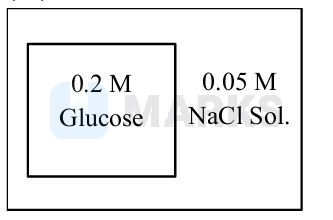
An artificial cell is made by encapsulating \(0.2 \mathrm{M}\) glucose solution within a semipermeable membrane. The osmotic pressure developed when the artificial cell is placed within a \(0.05 \mathrm{M}\) solution of \(\mathrm{NaCl}\) at \(300 \mathrm{~K}\) is _______ \(\times 10^{-1}\) bar. (nearest integer).
[Given : \(\mathrm{R}=0.083 \mathrm{~L} \mathrm{bar} \mathrm{mol}^{-1} \mathrm{~K}^{-1}\)]
Assume complete dissociation of \(\mathrm{NaCl}\)
[Given : \(\mathrm{R}=0.083 \mathrm{~L} \mathrm{bar} \mathrm{mol}^{-1} \mathrm{~K}^{-1}\)]
Assume complete dissociation of \(\mathrm{NaCl}\)
Solution:
2078 Upvotes
Verified Answer
The correct answer is:
25
$\begin{aligned} & \mathrm{NaCl} \longrightarrow \mathrm{Na}^{+}+\mathrm{Cl}^{-} \\ & 0.05 \mathrm{M} \quad 0.05 \mathrm{M} \quad 0.05 \mathrm{M} \\ & \text { Total } \mathrm{C}_1=0.05+0.05=0.1 \mathrm{M}(\mathrm{NaCl}) \\ & \mathrm{C}_2=0.2 \mathrm{M}(\text { glucose }) \\ & \pi=\left(\mathrm{C}_2-\mathrm{C}_1\right) \mathrm{RT} \\ & =(0.2-0.1) \times 0.083 \times 300 \\ & =2.49 \mathrm{bar} \\ & =24.9 \times 10^{-1} \text { bar }\end{aligned}$
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.