Search any question & find its solution
Question:
Answered & Verified by Expert
An element crystallises in a face–centred cubic (fcc) unit cell with cell edge a. The distance between the centres of two nearest octahedral voids in the crystal lattice is:
Solution:
1135 Upvotes
Verified Answer
The correct answer is:
In FCC octahedral voids are present at the edge centers and body center.
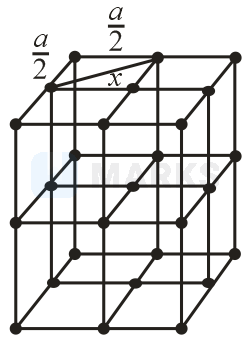
Minimum distance between centers of two octahedral voids
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.