Search any question & find its solution
Question:
Answered & Verified by Expert
An ideal gas expands isothermally from volume to and then it is adiabatically compressed back to its original volume . The initial and final pressures of the gas are and respectively and the net work done by the gas is , then
Options:
Solution:
2552 Upvotes
Verified Answer
The correct answer is:
The two processes are shown in the following P-V diagram:
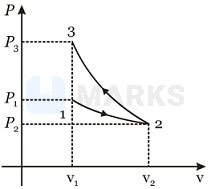
For iothermal process:
P1V1 = P2V2
i.e.,
For adiabatic process:
i.e.
As , hence P3 > P1
Further, as slope of adiabatic curve is greater than that of isothermal process curve, adiabatic curve will lie above the isothermal curve. That is, area under adiabatic curve > area under isothermal curve
i.e., Negative work > Positive work
i.e., W < 0

For iothermal process:
P1V1 = P2V2
i.e.,
For adiabatic process:
i.e.
As , hence P3 > P1
Further, as slope of adiabatic curve is greater than that of isothermal process curve, adiabatic curve will lie above the isothermal curve. That is, area under adiabatic curve > area under isothermal curve
i.e., Negative work > Positive work
i.e., W < 0
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.