Search any question & find its solution
Question:
Answered & Verified by Expert
An ideal gas is made to undergo a cycle depicted by the PV diagram alongside. The curved line from $\mathrm{A}$ to $\mathrm{B}$ is an adiabat.
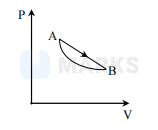
Then-
Options:

Then-
Solution:
1551 Upvotes
Verified Answer
The correct answer is:
Heat is absorbed in the upper part of the straight line path and released in the lower part
From the analysis of $\mathrm{P}-\mathrm{V}$ diagram we can easily say that $\mathrm{B}$ is the correct option.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.