Search any question & find its solution
Question:
Answered & Verified by Expert
An ideal gas is made to undergo the cyclic process shown in the figure below.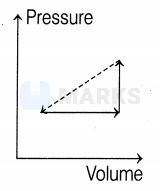
Let depict the work done, be the change in internal energy of the gas and be the heat added to the gas. Sign of each of these three quantities for the whole cycle will be ( refers to no change)
Solution:
1864 Upvotes
Verified Answer
The correct answer is:
Given cyclic process is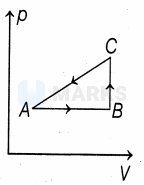
Area under compression process is more than area under expansion process . So, net work done is negative.
i.e.
Also, in a cyclic process, change in internal energy is zero.
i.e.
Now, by using first law of thermodynamics, we have
we see that,
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.