Search any question & find its solution
Question:
Answered & Verified by Expert
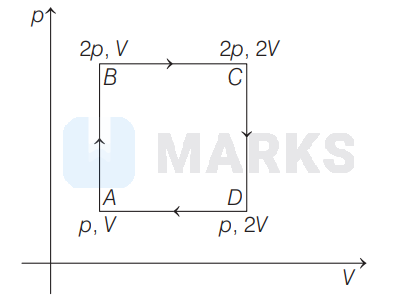
An ideal monoatomic gas is taken around the cycle $A B C D A$ as shown in the $p-V$ diagram. The work done during the cycle is given by
Options:

Solution:
2108 Upvotes
Verified Answer
The correct answer is:
$p V$
Work done in a cyclic process is equal to the area enclosed by the cycle in $p-V$ diagram.
$\begin{aligned}
\therefore \quad W &=\text { Area } A B C D \\
&=A B \times A D \\
&=(2 p-p) \times(2 V-V)=p \times V=p V
\end{aligned}$
$\begin{aligned}
\therefore \quad W &=\text { Area } A B C D \\
&=A B \times A D \\
&=(2 p-p) \times(2 V-V)=p \times V=p V
\end{aligned}$
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.