Search any question & find its solution
Question:
Answered & Verified by Expert
An ideal monoatomic gas undergoes an expansion from state to state following a process which is shown in the indicator diagram. If initially during the expansion, the gas was absorbing heat and later on it was rejecting heat, then what was the volume of the gas when it started rejecting the heat?
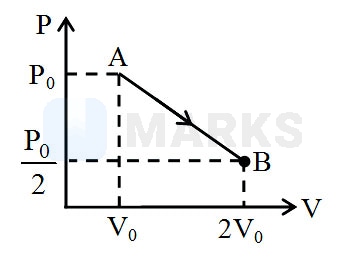
Solution:
1191 Upvotes
Verified Answer
The correct answer is:
From the graph we can find the pressure of the gas in terms of volume
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.