Search any question & find its solution
Question:
Answered & Verified by Expert
An octahedral complex with the formula \(\mathrm{CoCl}_3 \cdot \mathrm{nNH}_3\) upon reaction with excess of \(\mathrm{AgNO}_3\) solution gives 2 moles of \(\mathrm{AgCl}\). Consider the oxidation state of \(\mathrm{Co}\) in the complex is ' \(x\) '. The value of " \(x+n\) " is ______
Options:
Solution:
2843 Upvotes
Verified Answer
The correct answer is:
8
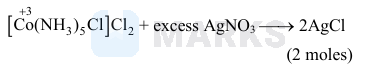
$\begin{aligned} & \mathrm{x}+0-1-2=0 \\ & \mathrm{x}=+3 \\ & \mathrm{n}=5 \\ & \therefore \mathrm{x}+\mathrm{n}=8\end{aligned}$
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.