Search any question & find its solution
Question:
Answered & Verified by Expert
Arrange the following alkyl halides in decreasing order of the rate of $\beta$-elimination reaction with alcoholic $\mathrm{KOH}$.
(a)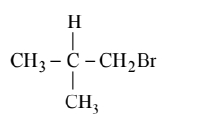
(b)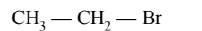
(C)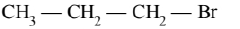
Options:
(a)
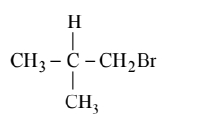
(b)
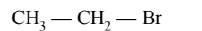
(C)
Solution:
1124 Upvotes
Verified Answer
The correct answer is:
$\mathrm{A}>\mathrm{C}>\mathrm{B}$
$\mathrm{A}>\mathrm{C}>\mathrm{B}$
Alkyl halides on heating with alcoholic $\mathrm{KOH}$ eliminates halogen from $\alpha$-carbon atom and hydrogen is eliminated from $\beta$-carbon atom to form an alkene.
(A)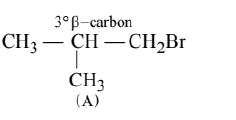
(B)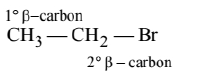
(C)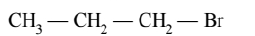
More the number of $\beta$-substituents (alkyl groups) more stable alkene will be formed on $\beta$-elimination, more will be the reactivity. Thus the correct order is $\mathrm{A}>\mathrm{C}>\mathrm{B}$
(A)
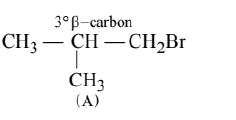
(B)
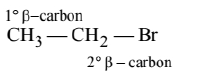
(C)
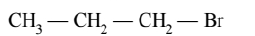
More the number of $\beta$-substituents (alkyl groups) more stable alkene will be formed on $\beta$-elimination, more will be the reactivity. Thus the correct order is $\mathrm{A}>\mathrm{C}>\mathrm{B}$
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.