Search any question & find its solution
Question:
Answered & Verified by Expert
Arrange the following in the correct order of their acidic strength
$\begin{array}{cccc}\mathrm{H}_2 \mathrm{C}=\mathrm{CH}_2 & \mathrm{CH} \equiv \mathrm{CH} & \mathrm{CH}_3-\mathrm{C} \equiv \mathrm{CH} & \mathrm{CH}_3-\mathrm{CH}_3 \\ \text { I } & \text { II } & \text { III } & \text { IV }\end{array}$
Options:
$\begin{array}{cccc}\mathrm{H}_2 \mathrm{C}=\mathrm{CH}_2 & \mathrm{CH} \equiv \mathrm{CH} & \mathrm{CH}_3-\mathrm{C} \equiv \mathrm{CH} & \mathrm{CH}_3-\mathrm{CH}_3 \\ \text { I } & \text { II } & \text { III } & \text { IV }\end{array}$
Solution:
1170 Upvotes
Verified Answer
The correct answer is:
IV $ < $ I $ < $ III $ < $ II
Acidic strength $\alpha \%$ s-character.
Due to maximum \% s-character of ethyne $(50 \%)$, hydrogen atoms attached to sp hybridised carbon of ethyne are most acidic followed by those attached to $\mathrm{sp}^2$ hybridised carbon of ethene $(33 \%)$ and those attached to $\mathrm{sp}^3$ hybridised carbon of ethane $(25 \%)$
In case of propyne, acidic character decreases than that of ethyne due to $+\mathrm{I}$ effect.
Hence, order of acidic strength is:
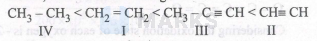
Due to maximum \% s-character of ethyne $(50 \%)$, hydrogen atoms attached to sp hybridised carbon of ethyne are most acidic followed by those attached to $\mathrm{sp}^2$ hybridised carbon of ethene $(33 \%)$ and those attached to $\mathrm{sp}^3$ hybridised carbon of ethane $(25 \%)$
In case of propyne, acidic character decreases than that of ethyne due to $+\mathrm{I}$ effect.
Hence, order of acidic strength is:
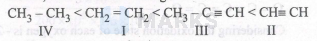
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.