Search any question & find its solution
Question:
Answered & Verified by Expert
As isolated box, equally partitioned contains two ideal gases $\mathrm{A}$ and $\mathrm{B}$ as shown
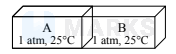
When the partition is removed, the gases mix. The changes in enthalpy $(\Delta \mathrm{H})$ and entropy $(\Delta \mathrm{S})$ in the process, respectively, are
Options:

When the partition is removed, the gases mix. The changes in enthalpy $(\Delta \mathrm{H})$ and entropy $(\Delta \mathrm{S})$ in the process, respectively, are
Solution:
2202 Upvotes
Verified Answer
The correct answer is:
zero, positive
According to KTG
Force of attraction and repulsion amongst molecules of ideal gas are negligible
So, $\Delta \mathrm{H}=0$
and randomness increases due to increase in volume so $\Delta \mathrm{S}=+\mathrm{ve}$.
Force of attraction and repulsion amongst molecules of ideal gas are negligible
So, $\Delta \mathrm{H}=0$
and randomness increases due to increase in volume so $\Delta \mathrm{S}=+\mathrm{ve}$.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.