Search any question & find its solution
Question:
Answered & Verified by Expert
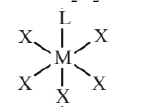
Assertion : Complexes of $\mathrm{MX}_6$ and $\mathrm{MX}_5 \mathrm{~L}$ type ( $\mathrm{X}$ and L are unidentate) do not show geometrical isomerism.
Reason : Geometrical isomerism is not shown by complexes of coordination number 6 .
[
Options:
Reason : Geometrical isomerism is not shown by complexes of coordination number 6 .
[
Solution:
1812 Upvotes
Verified Answer
The correct answer is:
If both Assertion and Reason are correct but Reason is not a correct explanation of the Assertion.
If both Assertion and Reason are correct but Reason is not a correct explanation of the Assertion.
Complexes of $\mathrm{MX}_6$ and $\mathrm{MX}_5 \mathrm{~L}$ type ( $\mathrm{X}$ and $\mathrm{L}$ are unidentate) do not show geometrical isomerism due to presence of plane of symmetry and necessary condition for showing geometrical isomerism is that complex is must of $\mathrm{MA}_4 \mathrm{~B}_2$ type or $\left[\mathrm{M}(\mathrm{AB})_2 \mathrm{X}_2\right]$ type

Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.