Search any question & find its solution
Question:
Answered & Verified by Expert
Assertion : Potassium ferrocyanide is diamagnetic whereas potassium ferricyanide is paramagnetic.
Reason : Crystal field splitting in ferrocyanide ion is greater than that of ferricyanide ion.
Options:
Reason : Crystal field splitting in ferrocyanide ion is greater than that of ferricyanide ion.
Solution:
2759 Upvotes
Verified Answer
The correct answer is:
If assertion is true but reason is false
Ferrocyanide ion :
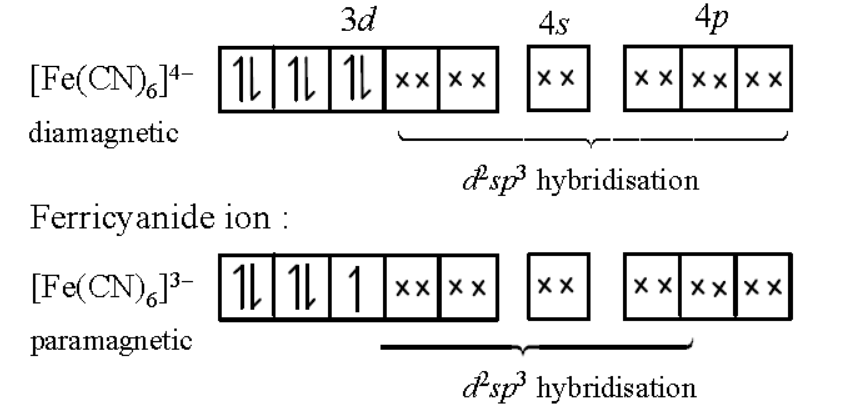
In ferrocyanide ion, the oxidation state of $\mathrm{Fe}$ is +2 however in ferricyanide ion, the oxidation number of $\mathrm{Fe}$ is +3 .
Generally, the higher the oxidation state of the metal, the greater the crystal field splitting. It means crystal field splitting in ferrocyanide ion is lower than that of ferricyanide ion.

In ferrocyanide ion, the oxidation state of $\mathrm{Fe}$ is +2 however in ferricyanide ion, the oxidation number of $\mathrm{Fe}$ is +3 .
Generally, the higher the oxidation state of the metal, the greater the crystal field splitting. It means crystal field splitting in ferrocyanide ion is lower than that of ferricyanide ion.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.