Search any question & find its solution
Question:
Answered & Verified by Expert
At constant pressure, the volume of a fixed mass of a gas varies as a function of temperature as shown in the graph.
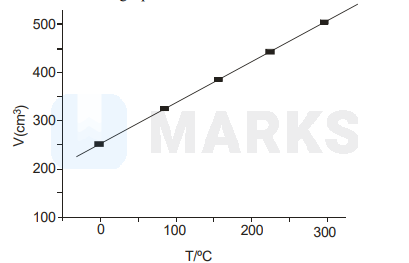
The volume of the gas at $300^{\circ} \mathrm{C}$ is larger than that at $0^{\circ} \mathrm{C}$ by a factor of
Options:

The volume of the gas at $300^{\circ} \mathrm{C}$ is larger than that at $0^{\circ} \mathrm{C}$ by a factor of
Solution:
1618 Upvotes
Verified Answer
The correct answer is:
2
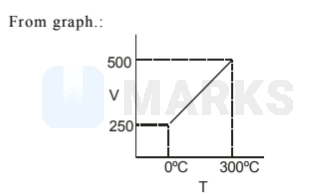
$\begin{array}{cl}\mathrm{T}_{1}=0^{\circ} \mathrm{C} & \mathrm{V}_{1}=250 \\ \mathrm{~T}_{2}=300^{\circ} & \mathrm{V}_{2}=500 \\ \frac{\mathrm{V}_{2}}{\mathrm{~V}_{1}}=2 & \end{array}$
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.